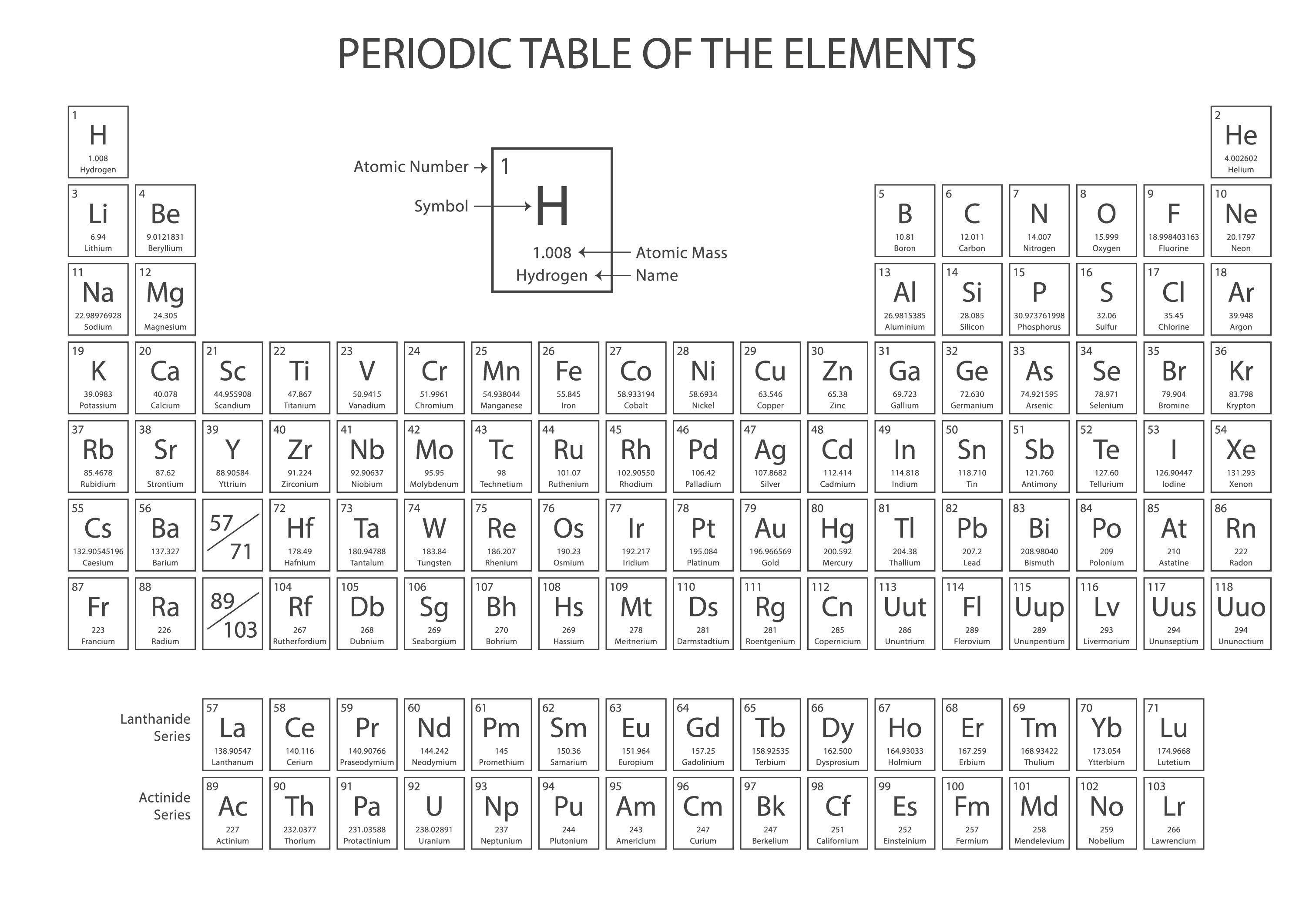
Elements in Group 18 of the periodic table are known as:ġ1. The chemical symbol “Na” represents which element in the periodic table?ġ0. Which element has the atomic number 1 and is the lightest element in the periodic table?ĩ. Elements in the same group of the periodic table have the same number of:Ī) Electrons in the outermost energy levelĨ. What are the columns of the periodic table known as?ħ. The rows of the periodic table are called:Ħ. What is the total number of known elements in the periodic table?ĥ. The atomic number of an element is determined by the number of:Ĥ. How are elements arranged in the periodic table?ģ. What is the organization of chemical elements based on their atomic number and recurring chemical properties called?Ģ. Part 1: 30 periodic table quiz questions & answersġ. 
Part 3: Free online quiz creator – OnlineExamMaker.Part 2: Download periodic table questions & answers for free.Part 1: 30 periodic table quiz questions & answers.They have partially filled d orbitals and often exhibit multiple oxidation states. Transition Metals: Groups 3 to 12 are known as transition metals. These elements have valence electrons in the s and p orbitals and exhibit a wide range of chemical behaviors. Main Group Elements: Groups 1, 2, and 13 to 18 are known as the main group elements or representative elements. Elements within the same group share similar chemical properties because they have the same number of valence electrons (electrons in the outermost energy level). Groups: The columns of the periodic table are called groups or families. The number of the period indicates the highest energy level that is partially filled with electrons.Ĭreate an auto-grading quiz/assessment without any coding – try OnlineExamMaker today! Each period corresponds to the energy levels (shells) occupied by electrons in the atoms of elements in that row. Periods: The rows of the periodic table are called periods. It is usually listed beneath the element’s symbol in the periodic table. For example, “H” represents hydrogen, “C” represents carbon, and “O” represents oxygen.Ītomic Mass: The atomic mass of an element is the weighted average mass of its naturally occurring isotopes. As of the latest knowledge, there are 118 known elements, ranging from hydrogen (the lightest) to oganesson (the heaviest).Ītomic Number: Each element in the periodic table is assigned a unique atomic number, represented by the symbol “Z.” The atomic number corresponds to the number of protons in the nucleus of an atom of that element.Ītomic Symbol: Each element is represented by a one- or two-letter chemical symbol. Key features and components of the periodic table:Įlements: The periodic table lists all known chemical elements, which are substances composed of atoms with the same number of protons in their atomic nuclei. The periodic table allows scientists to predict the properties of elements, study their relationships, and identify trends in their chemical behavior. It is one of the most important tools in chemistry as it provides a systematic way to categorize and understand the behavior of elements. The periodic table is a tabular arrangement of chemical elements, organized based on their atomic number, electron configuration, and recurring chemical properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
